Stains, Impurities, and Foreign Substances
The plastic material is impure or contaminated.
2025-11-07
| 23.1 Causes | 23.2 Solutions |
|---|---|
| 23.1.1 Raw materials, pellets, or pigments are impure. | 23.2.1 Replace the raw materials, dyes, or pigments. |
| 23.1.2 The plastic has decomposed or deteriorated. | 23.2.2 Adjust the processing conditions to prevent material decomposition. |
| 23.1.3 The barrel and hopper are not clean. | 23.2.3 Clean the barrel, hopper, and feeding tube. |
| 23.1.4 Foreign substances mixed in during preheating. | 23.2.4 Prevent foreign substances from entering during drying or material storage. |
2025-11-07
Whitening (Stress Whitening):
During demolding, excessive ejection force is applied to the surface areas around ejector pins or grooves. Due to forced demolding, high internal stress develops in these regions, sometimes causing deformation. Whitening is a sign of stress cracking. Materials such as ABS and HIPS are particularly prone to this phenomenon.
2025-11-07
Dull Surface (Lack of Gloss):
Due to poor mold surface finish, inadequate contact between the molten material and the mold surface, or improper material and mold temperatures, the molded product fails to exhibit the natural gloss of the resin. Instead, the surface appears whitish, cloudy, or dull.
| 21.1 Causes | 21.2 Solutions |
|---|---|
| 21.1.1 Poor mold surface finish; presence of scratches, moisture, or oil contamination. | 21.2.1 Repair or clean the mold surface and wipe the cavity dry. |
| 21.1.2 Excessive or improper use of mold release agent. | 21.2.2 Choose a suitable release agent, apply sparingly, and wipe the cavity if necessary. |
| 21.1.3 Moisture or excessive volatiles in the plastic. | 21.2.3 Dry the material according to process requirements; if necessary, increase drying temperature or extend drying time. |
| 21.1.4 Decomposition or deterioration of plastic or pigment. | 21.2.4 Replace with qualified material. |
| 21.1.5 Poor plastic flowability. | 21.2.5 Use a material with better flowability or increase mold temperature and injection pressure appropriately. |
| 21.1.6 Low material temperature, low mold temperature, or slow injection speed. | 21.2.6 Increase material temperature, mold temperature, and injection speed. |
| 21.1.7 Excessive air in the melt or poor mold venting. | 21.2.7 Increase back pressure appropriately and add venting slots in suitable mold locations. |
| 21.1.8 Injection speed too fast and small gate size causing melt vaporization, resulting in a milky thin layer. | 21.2.8 Reduce injection speed and enlarge gate size to prevent melt vaporization. |
| 21.1.9 Material and mold temperatures too high or too low (each plastic type has an optimal temperature range for gloss; deviations reduce gloss). | 21.2.9 Adjust material and mold temperatures according to the specific resin requirements. |
| 21.1.10 Insufficient feeding or poor plasticization. | 21.2.10 Adjust feed volume to ensure uniform plasticization. |
| 21.1.11 Mixed or incompatible materials. | 21.2.11 Replace with qualified material. |
| 21.1.12 Insufficient draft angle causing poor demolding. | 21.2.12 Increase draft angle to ensure easy ejection of parts. |
| 21.1.13 Low surface hardness of molded parts, prone to scratching and abrasion. | 21.2.13 Design and choose proper packaging to protect product surfaces from friction. |
| 21.1.14 Uneven or oversized pellets. | 21.2.14 Use uniform pellets or adjust processing conditions for even plasticization. |
| 21.1.15 Exposed filler or non-oriented silver flake filler. | 21.2.15 Increase mold temperature to ensure filler is not exposed. |
| 21.1.16 Presence of silver streaks or uneven coloration. | 21.2.16 Ensure proper drying of material, identify cause of color unevenness, and take corrective measures. |
| 21.1.17 Hot runner temperature too high or too low; heater or thermocouple damaged. | 21.2.17 Adjust hot runner temperature or replace heater and thermocouple. |
2025-10-29
Titanium dioxide has the characteristics of high chemical stability, non toxicity, and good photoelectric performance, especially rutile titanium dioxide has high surface activity, which is very suitable for battery material modification. Like polyethylene glycol, the introduction of titanium dioxide is also to compensate for the insufficient energy density and rate performance of lithium iron phosphate itself.

There are three main ways to add titanium dioxide to lithium iron phosphate:
1、 Doping modification. By incorporating nanoscale titanium dioxide particles into the lattice of lithium iron phosphate, a heterostructure can be formed, significantly improving the conductivity of the material. Experiments have shown that doping 1% titanium dioxide can increase the electronic conductivity of lithium iron phosphate by two orders of magnitude and improve rate performance by 15% to 30%.
2、 Surface coating. Titanium dioxide can form a protective film on the surface of lithium iron phosphate particles, reducing direct contact between the particles and the electrolyte, thereby suppressing side reactions such as iron leaching and excessive growth of SEI film, while enhancing electrode water retention capacity and improving battery performance in low humidity environments.
3、 Photocatalytic assisted synthesis. Under ultraviolet light, titanium dioxide generates electron hole pairs, accelerating the redox reaction of the precursor, shortening the hydrothermal synthesis time, and improving the purity of the product. A study has successfully reduced the hydrothermal reaction time from 12 hours to 8 hours.
From a mechanistic perspective, the introduction of titanium dioxide can adjust the band structure of lithium iron phosphate, narrow the band gap, and enhance its response to visible light; Heterojunction interfaces can accelerate electron transfer.
In addition, the rigid structure of titanium dioxide can also buffer volume changes during charge and discharge processes, improving cycle life - for example, the capacity retention rate can still exceed 90% after 2000 cycles.
In practical industrialization, titanium dioxide and polyethylene glycol are often used in synergy. For example, the two together form a "carbon titanium dioxide" double coating structure, which not only improves conductivity but also enhances interface stability, resulting in a first discharge specific capacity of 165mAh/g for lithium iron phosphate, with a capacity decay of less than 5% after 1000 cycles.
But currently, the cost of nano titanium dioxide is relatively high, and the uniformity of dispersion is also a technical difficulty, which requires further optimization through processes such as ultrasound and ball milling. Looking ahead, optimizing process parameters through machine learning, developing low-cost synthesis methods for titanium dioxide, and constructing a ternary composite system of polyethylene glycol titanium dioxide graphene will be important research directions. In addition, efficient recycling technologies for waste lithium iron phosphate batteries containing titanium dioxide also need to be developed.
In summary, titanium dioxide significantly enhances the electrochemical performance and stability of lithium iron phosphate materials through various methods such as doping, coating, and photocatalysis, providing solid support for the further development of power batteries and energy storage systems.
SAT NANO is a best supplier of titanium dioxide powder in China, we can supply 30nm, 50nm, 100nm particle, if you have any enquiry, please feel free to contact us at admin@satnano.com
2025-10-29
The meanings of metamorphic treatment, grain refinement, and primary phase refinement or morphology change are different, and sometimes the term metamorphic treatment is used to summarize these contents.

(1) Deterioration treatment. For simple binary Al Si alloys, such as Z102 containing 11%~13% Si, it is a typical eutectic alloy. Its organization consists of coarse needle shaped (should be layered) silicon and a eutectic of alpha (Al) solid solution, as well as a small amount of block shaped primary silicon (with alloy composition biased towards the upper limit). The mechanical properties of the alloy are not high, with a tensile strength not exceeding 140 MPa and an elongation of less than 3%. If a modifier (2-3%) containing Na or NaF is added to the melt before pouring for modification treatment, it seems that the eutectic point is shifted to the right and the eutectic temperature is lowered after modification treatment, causing the originally slightly hypereutectic alloy to become a hypoeutectic alloy. The primary silicon disappears, the eutectic is refined, and a primary α (Al) solid solution appears. The mechanical properties of the alloy have been improved, with a tensile strength of 180 MPa and an elongation of over 8%. It can even be pressure processed into plates and strips.
Using sodium salt as a modifier is prone to moisture absorption, difficult to store, and difficult to control the amount of sodium added. Castings are prone to porosity, and the duration of the modification effect is short. After the modification treatment is completed, it must be poured within 30 minutes. Later, it was discovered that not only sodium, but also strontium had the same effect and had a long shelf life, known as long-acting spoilage agents. Therefore, using sodium salt (NaF) or Al Sr intermediate alloy, it is obvious that Al Sr intermediate alloy is superior, easy to store, easy to control the amount added, and has a long shelf life.
The effect of adding phosphorus is opposite to that of sodium. Originally a eutectic alloy, it obtains a hypereutectic structure, which increases the amount of bulk primary silicon and improves the wear resistance of the alloy. The eutectic cast aluminum alloy used for producing pistons should be treated with phosphorus modification.
Using antimony for modification treatment does not show significant changes in the alloy casting structure, similar to the unmodified structure. However, after solid solution aging, the structure undergoes significant changes, especially the strength and toughness of the alloy are significantly improved.
(2) Refine grain size. Adding a small amount of titanium to aluminum alloy to refine the alpha (Al) grains can improve both the mechanical properties and processability of the alloy. The process of refining the structure of aluminum alloy by adding titanium is also known as modification treatment.
Grain refining agents must be nucleation promoters. When metals and alloys solidify into crystals, they undergo nucleation and growth processes. The formation of crystal nuclei can be divided into homogeneous nucleation and heterogeneous nucleation. Homogeneous nucleation must ensure that the entire melt has the same degree of undercooling. In actual production, it is impossible to achieve this. There are always places (such as the mold wall) that cool quickly, and some places (such as the center of the casting) that cool slowly, resulting in different degrees of undercooling and varying nucleation times. During production, it is expected to add a substance that can be used as a crystal nucleus to achieve a uniform and fine structure in the casting. Adding a small amount of titanium or a small amount of titanium and boron to aluminum alloy can achieve this goal. In addition, cooling rate is also an important factor, which not only affects nucleation but also controls the growth rate of crystals. Only by considering both nucleation (including external crystal nuclei) and cooling rate can a uniform and fine microstructure be obtained.
(3) Change the initial appearance. There are often primary needle like (flake like) compound phases in aluminum alloys, which have a significant impact on the alloy's properties, especially its mechanical properties. If Al3Fe is a needle like phase, the mechanical properties will decrease. If Fe: Si=4:1, AFeSi phase is formed, which changes the shape and significantly improves the mechanical properties, especially elongation. Adding manganese can also alter the morphology of iron containing phases.
In AI Mg alloys, when the magnesium content is high, the β (Mg5Al8) phase becomes coarse and exists at grain boundaries. Without solid solution treatment, the mechanical and corrosion resistance properties are poor. However, solid solution treatment requires a long time. If a small amount of zirconium is added, the β phase can be refined, which is beneficial for heat treatment.
In aluminum alloys, there are occasionally impurities of low melting point metals (such as Pb, Sn, Bi, etc.) present, which weaken the grain boundaries and cause melting at slightly higher temperatures; For example, aluminum contains sodium, causing "sodium embrittlement". Adding a small amount of rare earth elements to turn these metal impurities into compounds can eliminate the problems present in grain boundaries.
Therefore, it can be said that in the process of melting and casting, the elements or intermediate alloys that can improve the alloy structure, refine the alloy grains, change the size, morphology or distribution of primary phases, and remove harmful phases at grain boundaries are all called modifiers, and this process can be called modification treatment.
SAT NANO is a best supplier of aluminum silicon alloy powder AlSi particle in China, we can offer different mircon particle size, if you have any enquiry, please feel free to contact us at admin@satnano.com
2025-10-29
The demand for conductive paste in the photovoltaic industry and electronic packaging field is undergoing a transformation from high silver content to low silver or even silver free. Although silver powder has excellent conductivity and chemical stability, it is expensive, has limited resources, and is prone to electromigration. In contrast, copper's conductivity is second only to silver and its cost is about 1/100 of silver. Therefore, using low-cost copper powder instead of silver powder has become an important way to reduce costs. However, the surface of copper powder is prone to oxidation to form a thin layer of electrically insulating oxide, resulting in a serious decrease in conductivity and reliability. This makes preventing copper powder oxidation a core technical challenge in achieving the replacement of silver paste with copper paste.
Oxidation mechanism and performance degradation of copper powder
Copper is different from metals such as aluminum and nickel in that it is difficult to form a dense and stable intrinsic passivation layer on its surface. Therefore, the exposed copper surface will be continuously oxidized and corroded by oxygen and water vapor in the air. The smaller the particle size and larger the specific surface area of copper powder, the easier it is to rapidly oxidize to produce products such as cuprous oxide (Cu ₂ O) and copper oxide (CuO). This oxide insulation layer significantly reduces the conductivity of copper powder and hinders particle sintering connection, resulting in degradation of the performance of the conductive paste. Especially during the sintering process of the front electrode of photovoltaic cells (often requiring high temperatures exceeding 500 ℃), if copper powder is not protected, it will be severely oxidized and unable to form a good metal conductive network. In addition, in high temperature and high humidity environments, the growth of oxide layer can also cause the conductivity to deteriorate over time, affecting the lifespan of the device. Therefore, inhibiting surface oxidation of copper powder is crucial for maintaining its conductivity, sintering activity, and long-term stability.
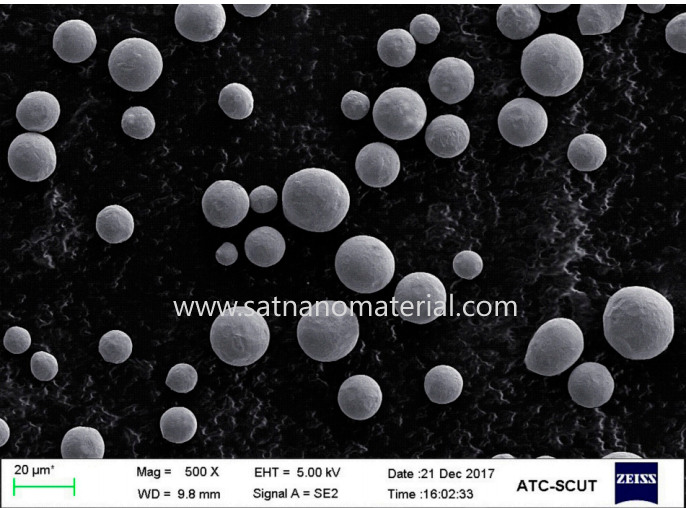
The main methods for surface antioxidant treatment of copper powder
Researchers and engineers have developed various surface antioxidant treatment techniques to address the issue of copper powder being prone to oxidation. Constructing a physical or chemical protective layer on the surface of copper powder can block oxygen contact or passivate active sites, thereby slowing down or even preventing oxidation from occurring. The main methods include organic coating protection, inorganic coating, self passivation alloying modification, and surface reduction passivation treatment. The following text introduces the principles and typical developments of each method separately.
Organic coating protection
Fatty acid and polymer coating: Long chain fatty acids such as oleic acid and stearic acid can bond with copper surfaces through carboxyl groups, forming a hydrophobic organic layer to isolate oxygen and moisture. Soaking copper powder in an oleic acid acetone solution for surface modification treatment can form a protective film of oleate on the surface of the powder. Experiments have shown that the purity and antioxidant properties of fine copper powder treated with this method are improved. Similarly, resins or polymers can also be used to coat the surface of copper powder. For example, the in-situ polymerization of polyaniline on the surface of copper powder to form a conductive polymer coating can effectively improve the antioxidant storage stability of copper powder in air. In addition, adding a certain amount of polymer binder (such as ethyl cellulose, acrylic resin, etc.) to pre coat copper powder during the preparation of conductive adhesive has also been proven to reduce the oxidation of copper powder and extend the shelf life of the slurry.
Silane coupling agent modification: One end of the organic silane coupling agent molecule contains a hydrolyzable silane group, which can react with copper surface oxides/hydroxyl groups to form silicon oxygen bonds, and the other end has an organic functional group to provide hydrophobic protection. The formation of an organic silicon film on the surface of copper powder through silane treatment can have a dual effect of anti-oxidation and improving dispersibility. Introducing silane coupling agent KH-902 into epoxy conductive adhesive to treat copper powder, the results showed that adding 3% significantly improved the oxidation resistance of copper powder during high-temperature curing process, and made the copper powder more evenly dispersed in the colloid. Research has shown that silane coupling agents can effectively inhibit the oxidation of copper powder below about 200 ℃, and their use in low-temperature curing slurry systems can help improve the compatibility and stability of the powder with organic carriers.
Inorganic coating
Precious metal coating: Coating a dense layer of inert metal on the surface of copper powder is an effective means of completely preventing oxidation. Silver coated copper powder has achieved mature technology and application. By chemical displacement plating or chemical reduction plating, silver shell layers of tens to hundreds of nanometers can be deposited on the surface of copper particles, forming a "copper core silver shell" structure. The silver shell layer isolates copper from the environment, greatly enhancing its antioxidant stability, while utilizing the high conductivity of silver to ensure excellent conductivity of the composite powder. Silver coated copper powder is expected to replace most pure silver powder and has been applied in fields such as conductive adhesives, electromagnetic shielding coatings, conductive inks, etc. It has also been used in photovoltaic slurries to achieve a silver reduction solution by reducing the silver content to 20% -50%. Another type is nickel plated copper powder, which can also act as a barrier layer to prevent copper oxidation. However, nickel has lower conductivity, and the thickness of the coating needs to be controlled to balance oxidation resistance and conductivity. In addition, copper thin wires coated with palladium/gold have been maturely applied in electronic packaging. For example, bonded copper wires are often coated with a layer of palladium and extremely thin gold to prevent copper from oxidizing and becoming brittle before high-temperature secondary bonding. Overall, the precious metal coating method has the best antioxidant effect, but still introduces a certain amount of precious metals, resulting in higher costs and precise control of coating thickness.
The application prospects of copper powder in conductive pastes and electronic packaging are broad, but oxidation has been the main obstacle between laboratory achievements and actual products. Recent studies have shown that various strategies such as organic coating, inorganic coating, self passivation alloying, and surface reduction passivation can significantly improve the antioxidant properties of copper powder, enabling it to maintain excellent conductivity within a wide process window. Different methods have their own advantages and disadvantages, and need to be selected or combined for specific applications.
SAT NANO is a best supplier of copper powder in China, we can offer 50nm, 100nm, 200nm, 500nm, 1-3um and 0-15um, if you have any enquiry, please feel free to contact us at admin@satnano.com
2025-10-29
First、 Physical Law
Physical methods can usually prepare copper powder with high purity and good sphericity.
1.Atomization method
Aerosolization method: using high-pressure inert gas, the resulting powder has low oxygen content and good sphericity, but the cost is relatively high. Commonly used in high-performance powder metallurgy and 3D printing of metal powders.
Water atomization method: using high-pressure water flow, fast cooling speed, irregular powder particles obtained (mostly in the form of flakes or tears), large specific surface area, and relatively high oxygen content. Low cost, commonly used for manufacturing diamond tools, friction materials, etc.
Principle: Molten copper liquid is sprayed out through a nozzle, crushed into small droplets using high-pressure gas (air, nitrogen, argon) or high-pressure water, and then condensed into spherical or nearly spherical powder under the action of surface tension.
Characteristics: High production efficiency, easy industrial large-scale production, and is one of the main methods for producing metal powders.
2.Vacuum evaporation condensation method
Principle: In a high vacuum environment, metal copper is heated and evaporated, and gaseous copper atoms are condensed into ultrafine powder on a low-temperature condensation wall.
Characteristics: It can prepare nanoscale, high-purity ultrafine copper powder, but the equipment is complex, the yield is low, and the cost is high. It is mainly used in laboratory research and high-precision fields.
Second, Chemical method
Chemical methods can prepare copper powder with finer particle size and diverse shapes, especially nano copper powder.
1.Liquid phase reduction method (the most commonly used chemical method)
Principle: Using soluble copper salts (such as copper sulfate and copper chloride) as raw materials and hydrazine hydrate, formaldehyde, sodium borohydride, ascorbic acid (vitamin C) or glucose as reducing agents, Cu ² ⁺ is reduced to Cu ⁰ atoms in the liquid phase and aggregated into powder.
Reaction example (using copper sulfate and hydrazine hydrate as examples):
2CuSO₄ + N₂H₄·H₂O + 4NaOH → 2Cu↓ + N₂↑ + 2Na₂SO₄ + 5H₂O
Characteristics: The operation is relatively simple, and the particle size and morphology of copper powder can be controlled by adjusting parameters such as reactant concentration, temperature, pH value, and additives (dispersants, protectants). It is the most commonly used method for laboratory and industrial production of ultrafine copper powder.
2.electrolysis method
Principle: Use copper plate as the anode and titanium or stainless steel plate as the cathode, and apply electricity in an electrolyte containing copper ions. The copper at the anode loses electrons and dissolves into Cu ² ⁺. Cu ² ⁺ obtains electrons at the cathode and is reduced to metallic copper, which is deposited in powder form on the cathode.
Characteristics: The obtained copper powder has high purity (up to 99.95% or more), dendritic structure, large specific surface area, and good compressibility. But the energy consumption is high and the cost is also high. It is an important method for producing copper powder for electrical materials.
3. Thermal decomposition method
Principle: Copper compounds (such as copper formate, basic copper carbonate, and carbonyl compounds of copper) undergo thermal decomposition at a specific temperature, producing copper powder and gas products. Characteristics: The powder has high purity, but the raw material cost is high and may produce harmful gases, making it less commonly used.
3、 Mechanical Law
ball milling
Principle: Metal copper blocks or copper shavings are placed together with grinding balls in a ball milling jar. Through the high-speed rotation or vibration of the ball mill, the grinding balls strongly impact, crush, and friction the copper material, causing it to repeatedly break and cold weld, ultimately refining it into powder
Features: Simple equipment, high output, low cost. But the powder shape is irregular, the particle size distribution is wide, it is easy to introduce impurities, and the energy consumption is high. Mainly used for producing flake copper powder (such as copper gold powder for coatings) or alloying copper with other elements.
The above are several mainstream methods for synthesizing copper powder, and there are other methods that can also be used to produce copper powder.
SAT NANO is a best supplier of copper powder in China, we can offer 50nm, 100nm, 500nm, 1-3um, 5um and so on, if you have any enquiry, please feel free to contact us at admin@satnano.com
2025-10-29
Behind the flourishing development of many industries such as electronics and new energy, there is a seemingly insignificant but crucial material - conductive silver paste. Keyboards, mobile phones, tablets, solar panels, smart cards, RFID and other devices rely on them to achieve connections and fully function.
Silver has the highest conductivity among metals, with excellent conductivity, thermal conductivity, good chemical stability, and weldability, and is widely used in various fields of modern electronics industry. Whether it is an OLED flexible screen or an LCD screen, there are countless thin conductive lines inside the screen, which are printed with "conductive silver paste".

1. Composition of conductive silver paste
Conductive silver paste is usually a viscous paste prepared by mechanical mixing of micrometer sized metal silver particles, polymer binders, solvents, additives, etc.
Among them, silver powder is a conductive functional phase, and its intrinsic characteristics such as microstructure, morphology, size and distribution, surface activity, etc. have a significant impact on the conductivity of the final film layer.
Adhesive phase is the film-forming substance of conductive paste, which can be divided into two categories: inorganic adhesive phase and organic adhesive phase. Its main functions are twofold: firstly, it acts as a skeleton, bonding silver powder together to achieve the conductive function of wiring; The second is to act between the film layer and the substrate to achieve the bonding between the conductive film layer and the substrate.
Solvent is also an essential component of conductive paste, playing a role in dissolving resin and adjusting the viscosity of the paste.
In addition, additives also play an important role in conductive pastes, mainly including thixotropes, dispersants, leveling agents, antioxidants, etc., which improve the coating performance of the paste and the physicochemical properties of the film layer.
2. Conductive silver paste classified by curing method
Conductive silver paste can be divided into three categories based on the curing method: sintered conductive silver paste, low-temperature cured conductive silver paste, and UV cured silver paste.
A. Sintered conductive silver paste
Sintered conductive paste is made by mixing silver powder as the conductive phase, glass powder as the bonding phase, organic solvents and other additives as the organic carrier. This type of conductive silver paste is sintered into a film at a sintering temperature generally>500 ℃.
Sintered conductive silver paste is mainly used for high-temperature insulation substrates such as ceramics and glass. This type of conductive silver paste is commonly used in electronic components such as potentiometers and thick film circuits that can withstand high temperatures, such as MLCC internal electrodes, photovoltaic cell electrode printing, and PCB local conductive paths.
B. Low temperature curing conductive silver paste
Low temperature cured conductive silver paste is composed of silver powder, polymer organic system, organic solvent, and additives. The curing temperature is generally less than 300 ℃, and good adhesion of circuit wires to the substrate can be obtained using screen printing technology.
The main characteristics of low-temperature curing conductive silver paste are low curing temperature, high bonding strength, and stable electrical performance. It is suitable for conductive and thermally conductive bonding in low-temperature curing welding applications, such as quartz crystals, infrared pyroelectric detectors, piezoelectric ceramics, potentiometers, flash lamps, as well as shielding, circuit repairs, etc; It can also be used for conductive bonding in the wireless instrument industry.
Low temperature cured conductive silver paste can also be used for printing on various flexible substrates, such as PET, PC, PEN, PI, paper, thermoplastic elastomers, etc.
C.UV cured silver paste
The bonding in UV cured silver paste is more complex compared to the previous two, and should at least have UV curing monomers, photosensitizers, and crosslinking agents.
Its curing mechanism is also relatively complex. The photosensitizer is excited by ultraviolet light to become an activating molecule, which induces the copolymerization reaction of photosensitive polymers and ultimately crosslinks to form a network structure.
Due to the fact that film formation does not require heating, UV cured silver paste is often used as a thermal sensor for bonding and conducting components that do not require heating and curing.
Conductive silver paste, as one of the indispensable basic materials in the electronics industry, its performance optimization and technological innovation will directly affect the progress and development of the entire industry. With the continuous updating and progress of applications, we look forward to the wider application of conductive silver paste in the future.
SAT NANO is one of the best supplier of silver powder in China, we can offer nano particle and micron particle for client to do condutivity paste, if you have any enquiry, please feel free to contact us admin@satnano.com
2025-10-29
The factors that affect the forming process mainly include the properties of the powder, the characteristics and effectiveness of additives, and the pressure, pressurization method, and pressurization speed during the pressing process. The properties of the powder mainly include the particle size, particle size distribution, particle shape, and moisture content of the powder.

First. The influence of powder properties on the pressing process
(1) The influence of the hardness and plasticity of metal powder itself. The hardness and plasticity of metal powder have a significant impact on the pressing process. Soft metal powder is easier to press than hard metal powder, which means that in order to obtain a certain density of compact, the pressing pressure required for soft metal powder is much smaller than that for hard metal powder. Soft metal powder deforms greatly during compression, increases the contact area between powders, and is easier to increase the compact density. Hard metal powders with poor plasticity must use forming agents during compression, otherwise they are prone to compression defects such as cracks.
(2) The influence of metal powder friction performance. Metal powder has a significant impact on the wear of molds, and the lifespan of molds is short when compressing hard metal powder.
(3) The influence of powder purity. The purity (chemical composition) of the powder has a certain impact on the pressing process. The higher the purity of the powder, the easier it is to press. When manufacturing high-density parts, the chemical composition of the powder has a significant impact on its formability, as impurities mostly exist in the form of oxides, and metal oxide powders are mostly hard and brittle and exist on the surface of the metal powder. During compression, the compression resistance of the powder increases, the compression performance deteriorates, and the elastic aftereffect of the billet increases. If lubricants or forming agents are not used to improve its compression performance, the resulting density and strength of the compression will inevitably decrease.
The oxygen content in metal powders exists in the form of compounds or surface adsorption, and sometimes also exists in the form of irreducible impurities. When the powder is not fully reduced or left for too long after reduction, the oxygen content will increase and the compression performance will deteriorate.
(4) The influence of powder particle size and particle size composition. When the particle size and particle size composition of the powder are different, the behavior during the pressing process is inconsistent. Generally speaking, the finer the powder, the poorer its flowability, and the more difficult it is to fill narrow and deep mold cavities, making it easier to form bridges. Due to the fine powder, its loose density is low, and the filling volume in the mold is large, requiring a larger mold cavity size. In this way, the movement distance of the die and the internal friction between the powders will increase during the pressing process, resulting in an increase in pressure loss and affecting the uniform distribution of the compact density. Compared with coarse powders of the same shape, fine powders have poorer compressibility and better formability, due to the increased contact area between the particles of the fine powder. For spherical powders, the particle size of the powder has almost no effect on the density within a medium or large pressure range.
(5) The influence of powder shape. The shape of the powder has the greatest impact on the filling of the mold cavity. Powder with a smooth and regular surface, close to a spherical shape, has good flowability and is easy to fill the mold cavity, resulting in a uniform density distribution of the compact; However, filling powders with complex shapes is difficult and prone to bridging phenomena, resulting in uneven density of the compact due to uneven powder loading. This is particularly important for automatic pressing, as the powders used in production are often irregularly shaped. In order to improve the flowability of the mixture, granulation treatment is often required.
(6) The influence of loose packing density of powder. When the loose density is small, the height of the mold and the length of the punch must be large. When pressing high-density blanks, if the size of the blank is long, the density distribution is prone to unevenness. However, when the loose packing density is low, the contact area of the powder increases during the pressing process, and the strength of the compact is high, which is its advantage.
Second. The influence of lubricants and forming agents on the pressing process
1) The influence of lubricants and forming agents on the pressing process
Different metal powders must use different lubricants or forming agents.
Has appropriate viscosity and good lubricity, and is easy to mix evenly with powder materials.
No chemical reaction occurs with powder materials, making it easy to eliminate and leaving no harmful impurities during pre firing or sintering.
The loose density and flowability of the mixed powder are not significantly affected. Except for special circumstances, its softening point should be high to prevent melting due to temperature rise during the mixing process.
There is no adverse effect on product performance and appearance after sintering.
(2) The dosage and effect of lubricants and forming agents
The amount of lubricant and forming agent added is related to the type and particle size of the powder, the pressing pressure, and the friction surface area, as well as their own materials. The amount of lubricant added is approximately proportional to the shape factor.
Third. The influence of suppression methods on the suppression process
During the compression process, two parameters need to be selected, one is the pressurization speed and the other is the holding time. (1) The impact of pressurization methods. Due to pressure loss during the pressing process, uneven density of the billet may occur. To reduce this phenomenon, bidirectional pressing, multi-directional pressing, or changing the die structure can be used. For parts with complex shapes, composite die punching can be used during forming.
(2) The impact of pressure holding time. During the pressing process, if the powder is maintained at a specific pressure for a certain period of time, it can often achieve very good results.
SAT NANO is a best supplier of metal powder in China, we can offer silver powder, copper powder, gold powder and alloy powder, if you have any enquiry, please feel free to contact us at admin@satnano.com
2025-10-27
Polyvinyl alcohol (PVA) is one of the most important and widely used water-soluble polymers in industrial applications. Its preparation process involves first polymerizing vinyl acetate (VAc) to form polyvinyl acetate (PVAc). The vinyl acetate groups (-OAc) on the PVAc are then converted to hydroxyl groups (-OH) through an alcoholysis (hydrolysis) reaction. Based on the degree of alcoholysis, PVA is divided into two major series: fully hydrolyzed and partially hydrolyzed.

Fully hydrolyzed 99 series PVA (such as ElephChem pva 2699, 2499, 2099, and 1799) refers to grades with a degree of hydrolysis of 99.0 mol% or higher. This extremely high degree of hydrolysis is the core prerequisite for these PVA grades to achieve high performance, strength, and water resistance. This blog will analyze, from four perspectives: molecular structure, grade differentiation, performance advantages, and key application areas, how fully hydrolyzed 99 Series PVA has become the cornerstone of "hardcore" materials such as high-performance fibers, specialty films, and durable adhesives.
1.Molecular Structure Determine Performance: The Mechanism and Effect of Complete Hydrolysis
1.1 Hydroxyl Density and Hydrogen Bonding Network Construction
In the fully hydrolyzed 99 Series, nearly all hydrophobic vinyl acetate groups on the molecular chain are replaced by hydrophilic hydroxyl groups. Hydroxyl groups (-OH) are extremely polar functional groups that form strong intramolecular and intermolecular hydrogen bonds, building a highly dense and stable three-dimensional network.
This dense hydrogen-bonding network contributes to two crucial molecular effects:
- High crystallinity: Strong hydrogen bonding forces enable PVA molecular chains to stack neatly and tightly, forming highly ordered crystalline regions. This increased crystallinity is the fundamental reason for the high tensile strength and high modulus of 99 Series PVA.
- Water Resistance: The dense hydrogen bond network makes it difficult for external water molecules to penetrate the crystals at room temperature and disrupt the connections between the molecular chains, effectively preventing PVA from dissolving. Therefore, 99 series PVA is essentially insoluble in water at room temperature and typically requires hot water above 90°C to fully dissolve and disperse. This ensures its structural stability in humid environments and aqueous systems.
1.2 Linear Correlation between Degree of Polymerization and Viscosity/Strength
Assuming a constant degree of hydrolysis (HD>99.0%), the differences between fully hydrolyzed 99 series PVA grades are primarily determined by the average degree of polymerization (DP) or average molecular weight (MW). DP is a key parameter that determines the rheological properties of polymer solutions and the mechanical properties of the final product.
The DP ladder of ElephChem 99 series grades (based on the average DP):
- Ultra-High DP (Polyvinyl Alcohol 2699): DP = 2600-3000. These grades have the longest molecular chains and the highest degree of chain entanglement. Its highest solution viscosity imparts exceptional cohesive strength and adhesion to the cured material, making it an ideal choice for manufacturing high-strength, high-modulus fibers and specialty high-viscosity adhesives.
- Medium-high degree of polymerization (Polyvinyl Alcohol 2499 / Polyvinyl Alcohol 2099): DP = 2,000-2,500. This grade offers a balanced viscosity and mechanical properties. It is the most widely used grade for sizing agents in the textile industry and for general-purpose, high-performance coatings and films.
- Medium-low degree of polymerization (Polyvinyl Alcohol 1799): DP = 1,700-1,800. Its relatively low solution viscosity facilitates its use in systems with high solids content or requiring rapid penetration. For example, precursors for polyvinyl butyral (PVB) require precise molecular weight control (e.g., 1799 for PVB, MW = 76,000-82,000) to ensure efficient acetalization and the quality of the resulting interlayer film.
2. Core Performance Advantages of the Fully Hydrolyzed PVA 99 Series
- Excellent Mechanical Properties (High Strength, High Modulus): High crystallinity gives PVA high tensile strength and modulus. Wet or dry-wet spinning yields high-strength, high-modulus PVA fibers with properties comparable to those of ultra-high-density polyethylene (UHMWPE). These fibers are a key raw material for replacing asbestos in cement reinforcement and ballistic materials.
- Excellent Gas Barrier Properties: PVA films, particularly those produced from the 99 series, offer one of the best barrier properties against gases like oxygen and nitrogen among known polymer materials. The highly hydrogen-bonded network within their molecular structure prevents gas permeation, making them ideal as high-performance barrier layers for oxygen-sensitive food and pharmaceutical packaging.
- Chemical and Oil Resistance: The 99 series PVA shows good resistance to solvents, oils, greases, and weak acids and bases because its molecules are very stable and it has few non-crystalline areas. This makes it useful for industrial coatings and special glues.
- Thermal stability: High crystallinity gives 99 series PVA a higher glass transition temperature (Tg) and melting temperature (Tm), improving the material's resistance to heat deformation and upper temperature limit.
3. Analysis of Key Industrial Applications of Fully Hydrolyzed 99 Series PVA
The unique properties of 99 Series PVA make it irreplaceable in multiple high-value-added sectors:
3.1 High-Strength High-Modulus PVA Fiber (HTHM PVA Fiber)
This is one of the most valuable end-products of 99 Series PVA. For example, the 1799 grade, with a DP of approximately 1750, achieves a high degree of molecular orientation through specialized spinning, heat treatment, and stretching processes.
- Applications: Used to replace asbestos and steel mesh in construction, it reinforces cement, mortar, and concrete, significantly improving the material's impact resistance, freeze-thaw resistance, and fatigue resistance. It is widely used in civil engineering structures such as highways, water conservancy projects, tunnel linings, and cement slabs.
3.2 Textile and Paper Industry
- Textile Warp Sizing: High-polymerization grades such as 2499 and 2699 provide an extremely tough and smooth size film, significantly improving the abrasion resistance and breaking strength of warp yarns during weaving. They are the preferred size for high-density, high-count fabrics (such as denim and premium cotton).
- Papermaking Surface Sizing Agent: As a surface sizing agent, the 99 series PVA forms a high-strength film on the surface of paper, significantly improving its surface strength, folding resistance, and printability. This is crucial for high-end coated paper and specialty functional papers (such as thermal paper and dust-free paper).
3.3 Polyvinyl Butyral (PVB) Precursor
PVB is a core material for automotive safety glass and architectural laminated glass. As an intermediate in the acetalization reaction, the quality of PVA directly determines the optical clarity, toughness, adhesion, and aging resistance of the final PVB film. Grades: 1799 specialty grades (such as SX-I/II/III) with a DP ≈ 1700-1850 are precisely designed to ensure ideal molecular structure and uniform dispersion during the subsequent acetalization reaction, meeting the stringent optical quality requirements of safety glass.
3.4 High-Performance Building Adhesives and Dry-Mix Mortars
In the construction industry, 99-series PVAs are used as high-performance additives to improve material durability and adhesion.
- Applications: As secondary dispersing binders and water-retaining agents in mortars and putty powders, their high bond strength and water resistance ensure the stability and durability of wall putties, tile adhesives, and other materials in humid and temperature-stable environments.
4. Conclusion: Future Outlook for Fully Hydrolyzed 99-Series PVA
99-series PVAs are a classic and promising branch of polymer materials science. By precisely controlling the degree of hydrolysis and polymerization, as demonstrated by ElephChem's grade system, the industry can develop specialized grades tailored to meet the demands of diverse and demanding applications.
From high-strength fibers that reinforce modern infrastructure, to PVB interlayer films that ensure safety, to environmentally friendly, high-performance coatings that enhance quality of life, the 99 series PVA, with its unparalleled strength, stability, and water resistance, continues to play a key role as a driver of high-performance, "hardcore" materials in the upgrading and sustainable development of the global manufacturing industry. As novel uses, like 3D printing and medical hydrogels, ask for better PVA, studies into improving and changing the 99 series PVA will likely increase. This will probably expand its value in industry and its market potential.
Website: www.elephchem.com
Whatsapp: (+)86 13851435272
E-mail: admin@elephchem.com
Categories:
- Bismaleimide Series2
- Cross-Linking agent / Vulcanizing Agent1
- Curing Agent1
- Engineering Plastic Pellets4
- Epoxy Resin2
- Ethylene-VinylAlcohol Copolymer(EVOH)1
- Fish Oil1
- Food Additives3
- Glucosamine1
- Heat-resistant modifier series1
- High Assay Quaternary Ammonium Compounds9
- Low Assay Quaternary Ammonium Compounds13
- Modified Polyvinyl Alcohol1
- Monomalemide Series2
- Other Surfactants/Catalysts8
- Plastic Random Packing1
- Plastic Structured Packing1
- Polyacrylamide1
- Polyurethane Resin2
- Polyvinyl Alcohol (PVA)2
- Power Coatings3
- Quaternary Ammonium Hydroxide4
- Special Quaternary Ammonium Compounds7
- TPU4
- Tertiary Amines1
- UV Ink1
- VAE Emulsion (Vinyl Acetate–ethylene Copolymer Emulsion)1
- aluminum paste1
- antiform2
- fire sleeve2
- resin2
Tags: